AP Physics: Whiteboarding
Students whiteboarded their answers to yesterday’s problems. There were a couple of meaty ones on the worksheet, so I had all groups whiteboard the same problem and then we had several consensus-building conversations.
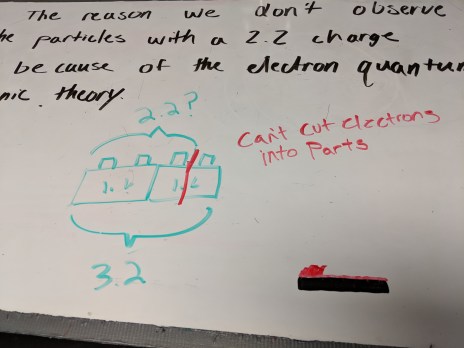
A visual for quantization of charge
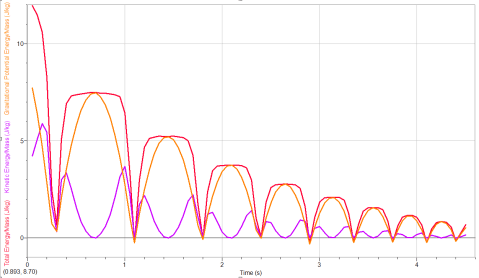
Physics: Bouncy Ball Energy
We wrapped up figuring out where a bouncy ball dissipates energy. As part of the follow-up, we dropped a kickball under a motion detector (thanks to Brian Frank for the idea!) to produce energy vs. time graphs and confirm the results from students’ video analysis.
Chemistry Essentials: Classifying Matter
Students worked on a worksheet to connect particle diagrams to the vocabulary for classifying matter that we introduced yesterday. I also was able to have really good individual interactions with a couple of students I’ve been butting heads with a lot, which helped make the whole classroom feel more relaxed and positive. I’ve been getting overwhelmed in chemistry a lot this year and have not done a good job of making time to talk to students about things besides chem, and I can’t underestimate the value of those conversations.




















