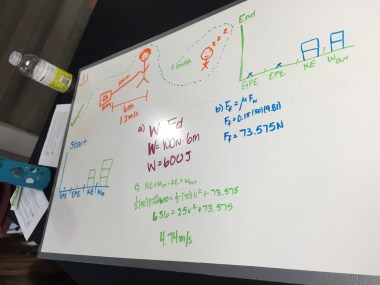
Physics: Ohm’s Law
Students wrapped up using the PhET circuit construction kit to develop Ohm’s Law. Students were able to pretty easily reason out the formula based on their graphs. Looking back, I wish I’d had students put their experimental conditions and equations on the boards, as well. I usually try to keep the boards pretty simple since I have some big classes, but I think that information would have added a lot of value to the conversation in this case.

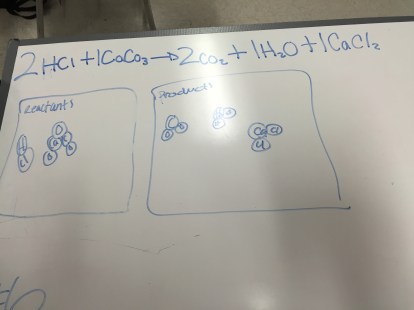
Chemistry: Limiting Reactant Practice
Today, students did some limiting reactant problems where the given and desired information is in grams, rather than moles. I was really pleased at how easily most of them worked through the problems. A lot of my students were struggling to connect ideas between different days earlier in the trimester, so it was great to see how many readily pulled out earlier skills and problem solving strategies to help today. I also saw a big jump in the quality of the questions I’m getting from my students; one student in particular was really focusing on the why when she was talking to me, when in the past she seemed most interested in getting something to write down. I’ve been having a lot of conversations with this class about how learning works and sharing why I do things the way I do, and I’m hoping I can get students to continue with the things I saw today.