Physics: Force Labs
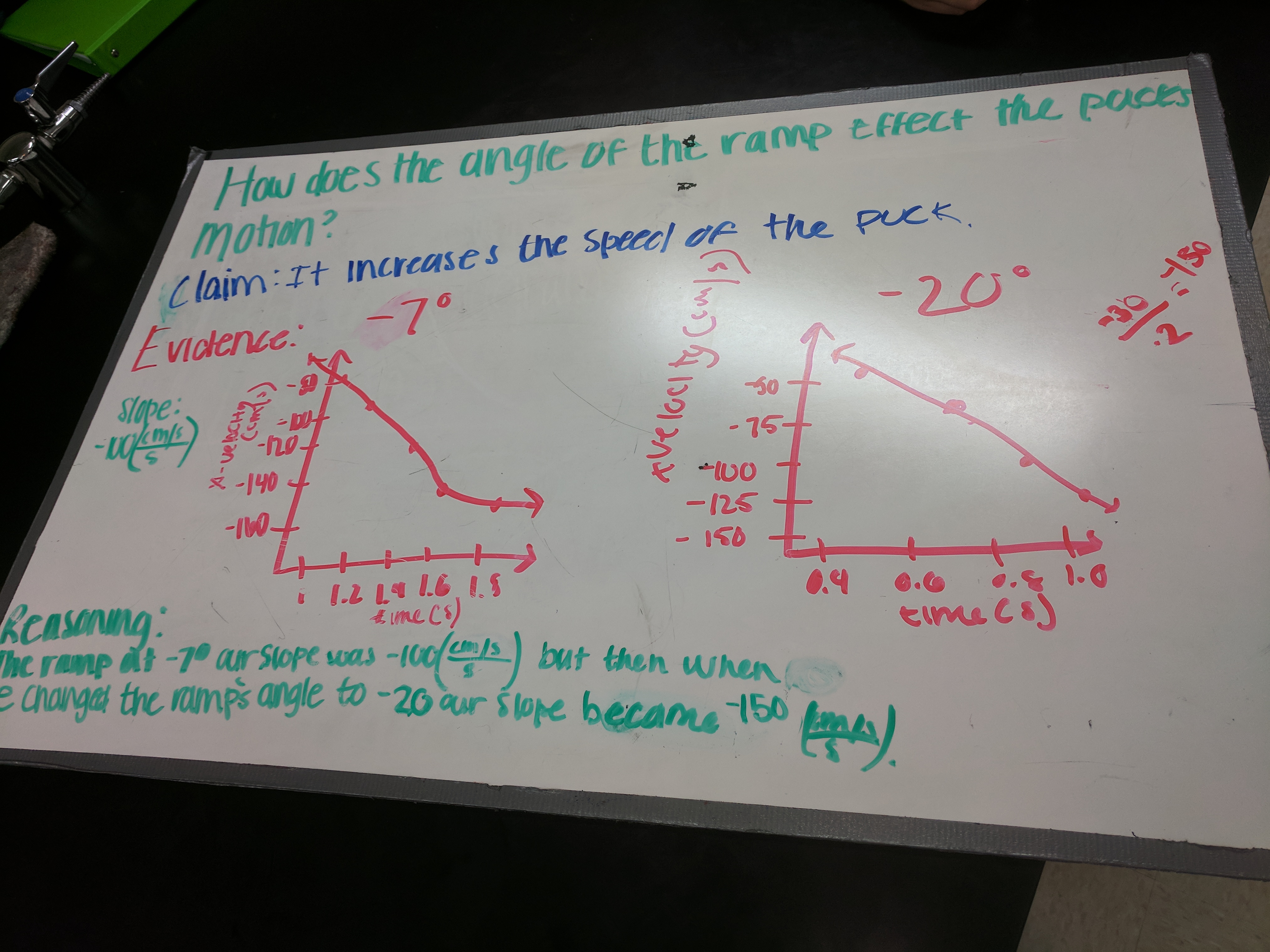
This week was the force of gravity lab and the spring force lab. Students have been struggling to draw meaning from labs, so I spent more time than usual on “translating” the line of best fit into physics and on the conclusions portion of our lab template for the gravity lab, which seemed to pay off. On the spring force lab, I tasked students with doing that more independently and encouraged students to use their force of gravity lab for reference. I noticed a lot of groups using Fg, rather than Fs, and N/kg as the slope unit, rather than N/cm, which tells me students were focusing on what the right answer was in the force of gravity lab, rather than why it was the right answer. This fits with some other observations I’ve made this year and tells me I need to keep thinking about how to get students focusing on their process in labs, rather than what should be the correct result.
The other physics teacher and I decided to use the spring force lab to introduce force sensors since we have some more involved labs coming up where they will need to use force sensors, and I was really pleased with how quickly students got comfortable with those as a tool. I think it was really valuable for students to have their first exposure one where the measurements were relatively straightforward. I tend to fall into using low-tech tools until there is a good reason to use probeware, but the downside is students are then learning to use the probeware in labs with relatively complex scenarios or when they need to use multiple probes to measure different things. I need to remember the value in starting to use probes in relatively simple labs where we don’t strictly need the probeware.
AP Physics 1: Impulse
This week we introduced momentum and impulse. We started with the Modeling Instruction cart explosion lab where students launch spring-loaded carts off each other and graph the ratio of the cart’s velocities vs. the ratio of the cart’s masses and establish the idea of momentum. Next, we did video analysis of two linked air pucks to introduce center of mass (based on an article in The Physics Teacher by Taylor Kaar, Linda Pollack, Michael Lerner, and Robert Engles). After that, we looked at the change in velocity of carts as they crashed into force sensors with hoop springs to introduce impulse. I’ve tried a few versions of that lab and have yet to have students collect satisfying data, so have been thinking about what I want to try instead. In the course of my thinking, I realized I don’t think I’m satisfied with the storyline of my unit. I like the cart explosion lab to introduce momentum and the center of mass piece to think about what it means to treat an object as a particle, but I think those activities lead more naturally into conservation of momentum than impulse. I think next year I want to try starting the momentum transfer model with the same two activities, but then go straight into conservation of momentum. One route to impulse from there could be asking what if we change the system, such as looking at just one cart at a time in the cart explosion lab to motivate new tools in our model.