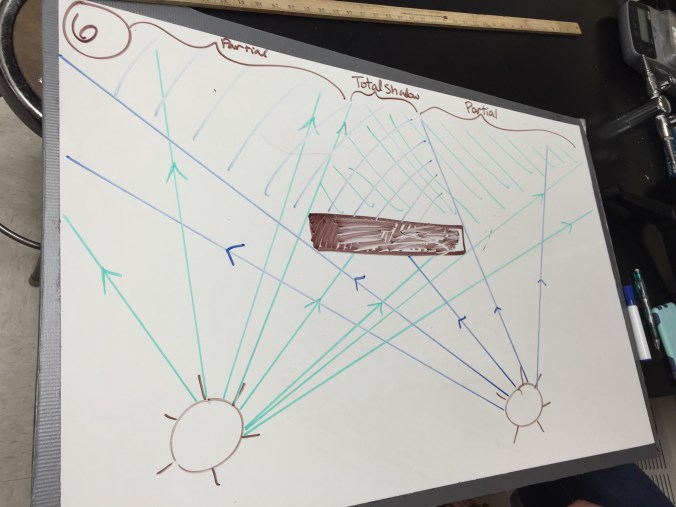
Physics: Curved Mirrors
We started by whiteboarding yesterday’s reflection problems. Along the way, I had a student stand in front of a nice, long horizontal mirror while someone else held a whiteboard between the student and the mirror to show a blocked object can still form an image. Afterward, students started making some qualitative observations about curved mirrors. My favorite part was when students noticed the hologram mirror I had out and start puzzling through what was going on. Even once the opened it up, students were eager to try changing the setup to see what would happen and test their ideas. I think some students were legitimately excited when I told them we’d draw the ray diagram for it later this week.

Chemistry: Neutralization Reactions
Students practiced determining the products in neutralization reaction. I was pretty pleased when a student asked “Aren’t these just double replacement reactions?” Last tri, a lot of students struggled to connect one day in class to the next, so this tri I’ve been much more intentional about trying to help students make those connections, so it’s gratifying to see students looking for links between new and old topics unprompted.