Today classes were shortened due to a pep fest.
AP Physics: KVL Diagrams
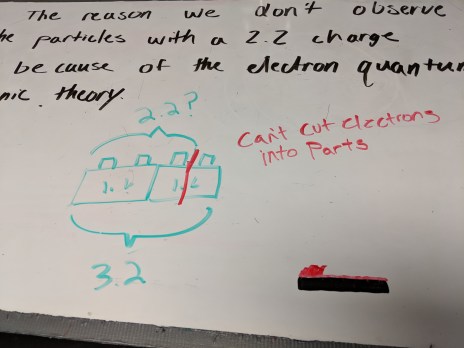
We had a very brief discussion about the results of the labs from Wednesday and Thursday; students were consistently very successful at picking up on they key patterns I wanted them to see. I also introduced them to Trevor Register’s KVL diagrams. I like to pair that with color-coded current paths on the circuit diagram.

Physics: TIPERs
Students worked through some conceptual problems on impulse and momentum, mostly taken from TIPERs to get them thinking about what the equations we have so far really mean. A lot of groups really wanted to start by guessing an answer, then come up with some physics to justify it, rather than the other way around. I’ve been encouraging students to use CER with these types of problem, starting with the evidence and working towards the claim, but I’m tempted to try and talk my department into switching to ERC to make the evidence first more explicit.
Chemistry Essentials: Formula Writing
Playing the mistakes game yesterday seemed to help some students start to make sense of writing chemical formulas, though a lot of students still need more practice. Today, I gave students some formulas and asked them to determine whether they are possible based on what we know about bonding. This seemed to help the concepts click for a few more students.