AP Physics: Projectile Video Analysis
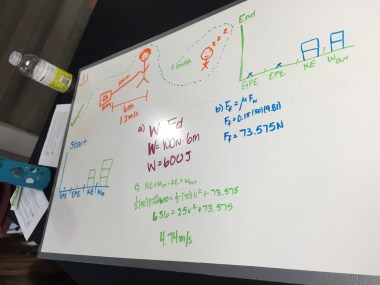
I gave students two videos of some students tossing a basketball and a video analysis guide I put together, then tasked them with getting the position vs. time and velocity vs. time graphs for each video of the basketball. I had a bunch of students who got genuinely excited when they looked at the velocity vs. time graphs and started putting together what they know so far to begin a model of projectile motion. One group even cheered when the slope of their y-velocity vs. time graph matched what their model so far!

Physical Science: Energy Skate Park
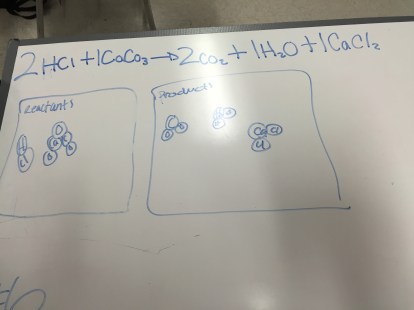
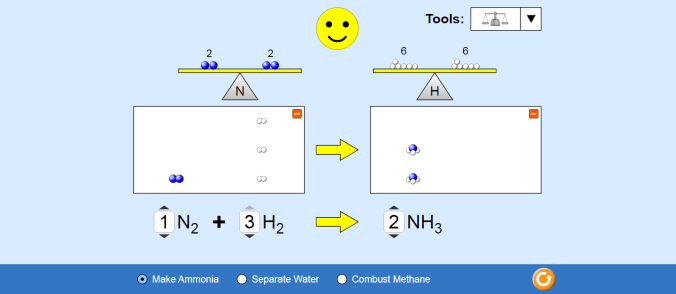
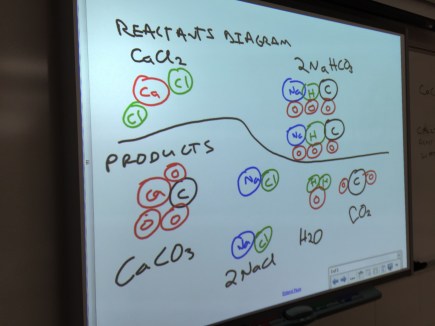
Students played with PhET’s Energy Skate Park simulation to look for how they could change the size of the various bars in the bar chart. Students pretty enthusiastically played with as many options as they could find, and made some nice observations, like how the motion of the skater changes when they switch to the moon or Jupiter.