AP Physics 1: Kirchoff’s Laws
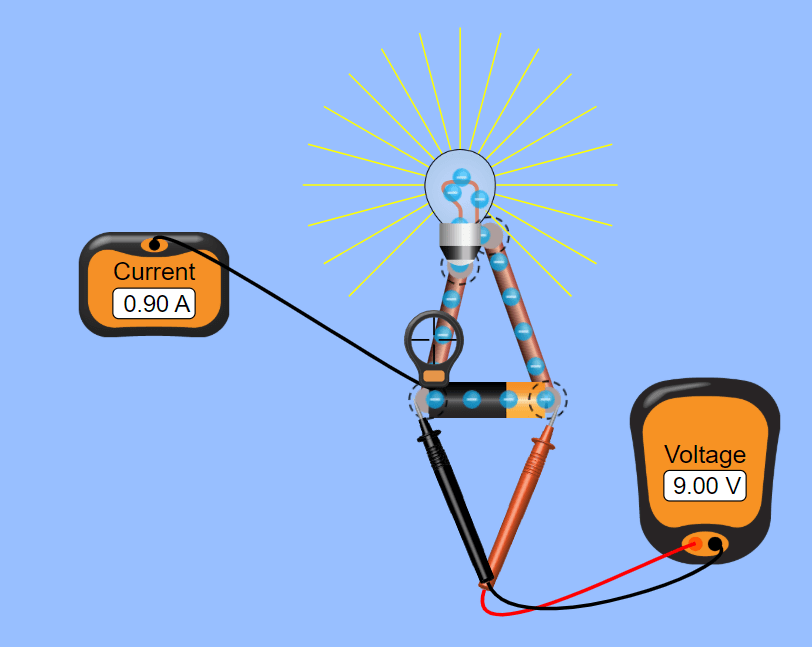
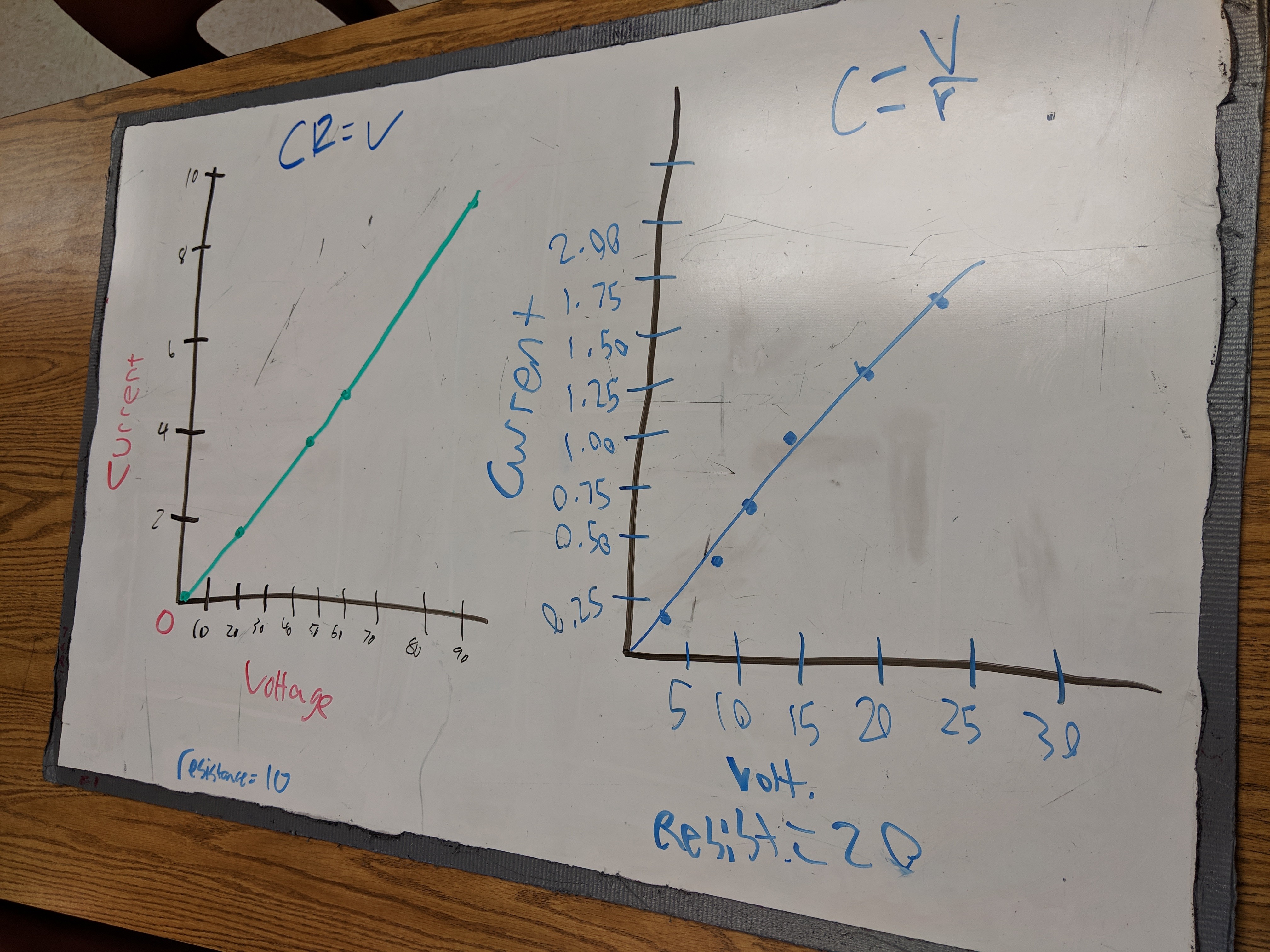
Students worked on developing Kirchoff’s Laws this week. We started with PhET’s circuit construction kit, then got out the power supplies and resistors. Some groups had trouble recognizing the simulation and the physical lab as addressing the same concepts, but explicitly asking groups how their results compared seemed to help students make the connections. There was also some good discussion about why the results in the physical lab didn’t match the simulation exactly. A thermal photo showed some heat at the alligator clips, which lead to some conversation about whether the wires we were using were ideal.

Physics: Energy Transfer Card Sort
This week we worked on starting energy conservation problems. To help the transition from bar charts to problems, I turned some problems Kelly O’Shea and Mark Schober wrote for the New Visions physics curriculum into a card sort. Seeing cards with two versions of the conservation of energy equation seemed to help a lot of students see how to build equations from the bar charts, which made the problems much smoother than in the past.