Physics: Energy Practical
Students were given an elastic string to hang from the ceiling. Students had to find k for their string, then figure out how high above the ground to hold an action figure attached to the end so that, when they dropped it, the figure would just barely hit the ground. In the past, I’ve used a bucket of water and we go by the sound (“splash, but no thud”). This year, the other physics teacher suggested using a force plate and looking for a small spike in the force vs. time graph. While students like the splashing, the force plate is a lot easier to move around, and makes it possible to capture slow-motion video.
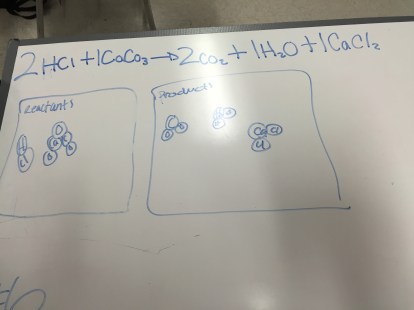
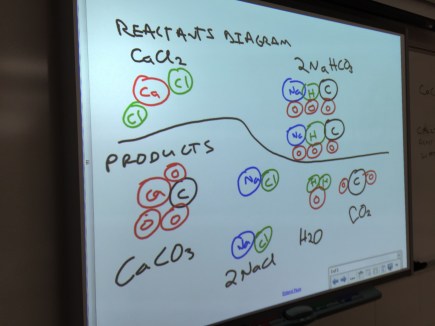
Chemistry: Mistakes Game
To go over Friday’s practice problems for balancing chemical equations, I had them do the mistakes game. Students focused on making their error an inconsistency in their representations of the reaction. I appreciated how willing my students were to try something new; they seemed to especially appreciate the safety inherent in the mistakes game.