Physics: Energy Introduction
To introduce energy, we used a lab my colleague has done in the past. Students dropped a steel ball into a bucket of sand and measure the depth of the crater. I haven’t told my students yet, but we’re using the crater depth as a proxy for energy, similar to the “chalk-smashing ability” in Etkina’s PUM curriculum. Students collected data and graphed to get a relationship between the crater depth and the drop height, which we’ll use tomorrow to help define gravitational potential energy. I also had students use free fall to calculate the impact speed of the ball and graph the relationship between crater depth and impact speed, which will lead to the definition of kinetic energy.

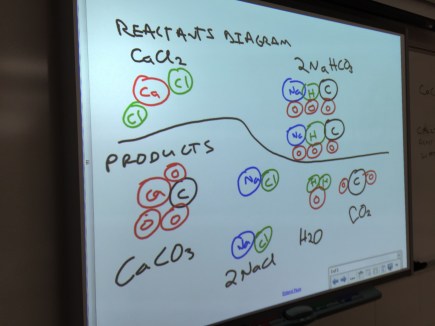
Chemistry: Conservation of Mass
This trimester, instead of teaching 9th grade physical science, I’m teaching the second half of a course called Chemistry Essentials. This two trimester course meets the minimum graduation requirements for my district. Most of the students tend to struggle in school, for a wide variety of reasons, and there is a high percentage of students who qualify for special education services.
The first unit deals with writing and balancing chemical equations, so I decided to start with conservation of mass. Students put calcium chloride, sodium bicarbonate, and bromothymol blue into a sealed plastic bag, tracking the mass of everything involved. Once the reaction was finished, they measured the mass of the bag and its contents and found the total mass had stayed the same. I also had them measure the mass after removing as much gas as possible. The main issue was students had some trouble getting the plastic bag all the way on the pan of the balance, which impacted the mass they measured at the end. Next time, I’ll try having students set their bag in a rigid container like a beaker to get the total mass.