Physics: Electric Fields
Students built off what they can see in the PhET Charges & Fields to develop the idea of what electric field lines represent. Students mostly got into trouble when they thought their answers were too obvious and tried to come up with something more complicated.

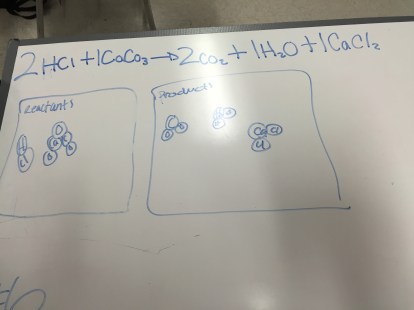
Chemistry: Endothermic & Exothermic Reactions
To introduce endothermic and exothermic reactions, we broke the terms down and talked about other words with the “therm” root and what that says about what these new terms might mean. Students then did some reading about endothermic and exothermic reactions. While the textbook doesn’t emphasize the role of energy in these reactions, I tried to bring that out in the short discussion after the reading.